Cubital tunnel Syndrome is a condition that occurs when there is pressure on the ulnar nerve at the elbow. This pressure can cause numbness and tingling in the fingers and hand. Symptoms can include numbness, tingling, and pain. Treatment options include surgery and/or physical therapy. Homeopathy is a system of medicine that is based on…
Read MoreBest Homeopathic Medicine for Epididymitis: The inflammation of the epididymis is called Epididymitis. The epididymis is a tube located at the back of the testicles that stores and carries sperm. Usually, the pain and swelling in the testicles are caused by the swollen epididymis. Men of all ages can affect by Epididymitis, but it’s most…
Read MoreBest Homeopathic Medicines for Ear Discharge: Otorrhea is another name for Ear discharge, which is any kind of fluid that comes from the ear. However, the natural oily secretions from your ear are called earwax. The purpose of earwax is to prevent ear viruses and bacteria. Sometimes ear infections can cause discharge ear. On the…
Read MoreBest Homeopathic Medicines for Dysuria: Dysuria is a symptom of pain, discomfort, or burning when urinating. It is more common in women than in men. In men, it is more common in older men than younger men. Homeopathic treatment for dysuria particularly aims in correcting the entire constitution of the patient. Homeopathic Medicines for dysuria…
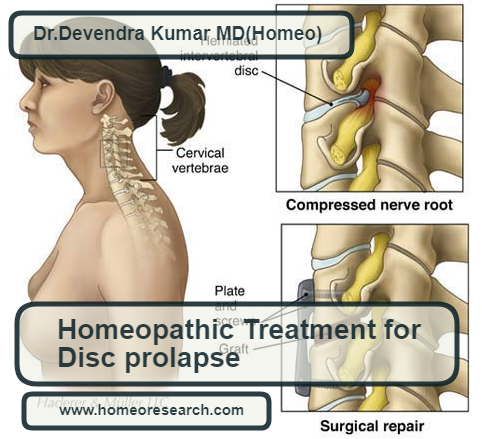
Read MoreBest Homeopathic medicines for disc prolapse: A spinal disc herniation is also sometimes called “disc prolapse”. Prolapse means “to fall out of place”, from the Latin probably meaning “to fall out”. The disc prolapse due to a problem with rubbery, cushion disc(s) in between the backbones. Even though Disc Prolapse is a local problem, Homeopathy…
Read MoreBest Homeopathic Medicine for Senile Dementia: Dementia describes a group of symptoms affecting memory, thinking, and social abilities severely enough to interfere with your daily life. It isn’t a specific disease, but several different diseases may cause dementia. Though dementia generally involves memory loss, memory loss has different causes. Having memory loss alone doesn’t mean…
Read MoreBest Homeopathic Medicines for Crohn’s Disease: Crohn’s disease is a type of inflammatory bowel disease (IBD). It causes inflammation of your digestive tract, which can lead to abdominal pain, severe diarrhea, fatigue, weight loss, and malnutrition. Inflammation caused by Crohn’s disease can involve different areas of the digestive tract in different people. In the case…
Read MoreBest Homeopathic Medicines for Claustrophobia: Claustrophobia is the fear of closed spaces. It can be caused by many situations or stimuli, including elevators, especially when crowded, windowless rooms, and rooms with closed doors and sealed windows. Even bedrooms with a lock on the outside, congested cars, and tight-necked clothing can induce a response in those…
Read MoreBest Homeopathic Medicine for Chalazion: A chalazion is a small swelling or lump on your eyelid because of a blocked gland. They’re called chalazia if you have more than one. A chalazion is one of the most common types of eyelid lumps. Homeopathic Medicines for Chalazion are most effective to cure it permanently. Moreover, there…
Read MoreBest Homeopathic Medicines for Carpal Tunnel Syndrome: Carpal tunnel syndrome (CTS) is a painful condition due to compression of the median nerve. As the nerve travels through the wrist at the carpal tunnel, inflammatory conditions can compress it. The main symptoms are pain, numbness, and tingling in the thumb, index finger, middle finger, and the…
Read More